Every material in the universe is believed to be made up of atoms. In an atom, electrons are bound to its nucleus by a strong force of attraction by the protons in the nuclei. In some materials few free electrons are available. These electrons can move freely along the material. Such materials with free electrons can conduct electricity.
When a charge (i.e., an excess or deficit of electrons) is applied to one side of such a conducting material, the electrons throughout will realign themselves, spreading out by virtue of their mutual repulsion, and thus conduct the charge to the other side.
Most of the conducting materials are metals. In metals, some electrons are always free for available for conduction. But the conductivity of these metals also depends on the amount of energy the electrons can transfer without colliding on the neighbouring atoms.
Conductivity
Conductivity is the reciprocal of resistance. When resistance is the opposition to the current flow, conductance is the amount of current that a material can conduct. Therefore, conductance can be defined as the ability of a material to conduct electric current. For example, a material with low resistance is highly conductive and vice Versa. Conductivity is denoted by the letter G.
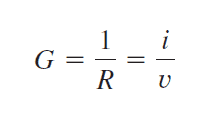
Unit of conductance
As conductance is the reciprocal of resistance it is mentioned in “mho” or “ Ω−1 ” or “℧”. Sometimes it is mentioned in “siemens” which is the derived unit of ℧ .

where Ω is the ohm, A is the ampere, and V is the volt.
Superconductors
Superconductivity is a property of a material to conduct without any resistance. Some material can conduct electricity at zero resistance when they are supercooled. For example, some ceramic metals can turn to superconductors when they are cooled to -319 deg. F. At this stage, the free electrons can move more easily losing their energy by collisions. Scientists are working on superconducting material and are finding ways how they can be used for electric power transmission.
